0
You have 0 items in your cart
RD Mathis Company Technical Library Publications
info@rdmathis.com | www.rdmathis.com

The usefulness of inert gases has steadily increased over the last few years, and with their use the requirements for purity have increased. In activities like sputtering, glove box processing, welding, crystal growing, gas chromatography, etc., there is a strong demand for a truly inert atmosphere, sometimes to be present for months at a time. While each application for an inert gas has its own special requirements, there are a number of common points that can be stressed.
It is the purpose of this article to consider purification methods relative to cost, convenience, efficiency, and ultimate purity. The article will be broken into three questions. First, what is an inert gas? Second, how is it purified? And finally, how can it be kept pure?
According to the Handbook of Chemistry and Physics[1], there are six inert gasses — helium (He), neon (Ne), argon (Ar), xenon (Xe), krypton (Kr), and radon (Rn). The inert gases are monatomic, and in general, do not chemically reacts[2, 3] with each other or with other elements. It is this chemical “inertness” property that makes these gases valuable as environments for highly reactive systems.
In contrast, other elemental gases — hydrogen (H), oxygen (O), and nitrogen (N) — react readily with other elements at easily available temperatures and only appear to be inert at room temperature because they form stable diatomic molecules H2, O2, and N2. Similarly, compound gases H2O, CO, CO2, NH3, hydrocarbons, etc. all react readily if broken down. The compound gases H2O, CO, etc. and. elemental gases, H2, O2, N2 shall be considered “reactive” gases for the rest of this article because they will be the primary impurities to be removed during purification for a truly inert environment.
Inert gases are obtained primarily by cryogenic fractional distillation. This process involves cooling the inert gas to a point just above its boiling point at a given pressure to remove all gases with boiling points above that of the inert gas, then liquefying the inert gas to pass off all of the impurity gases with boiling points below that of the inert gas. All of the inert gases can be fractionally distilled from the air. However, in the United States helium gas is distilled as a by-product from the liquification of natural gas in certain Texas wells.
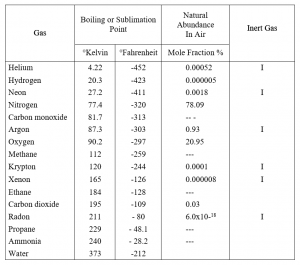
Table I shows the boiling points for the inert gases[1, 4], as well as other gases, and the natural abundance of inert gases in air[4]. The radioactive inert gas radon, primarily obtained from the radioactive decay of radium, is such a special case that it is only mentioned for completeness and will not be considered further.
TABLE I
GAS BOILING POINTS AND ABUNDANCE
Inert gases are primarily purified by three processes: a stringently controlled fractional distillation leading to bottled specialty gases, diffusion, and two forms of chem-adsorption[5, 6, 7, 8].
The first process, fractional distillation, has already been considered in the discussion on obtaining inert gases. The process can be cycled an infinite number of times. Consequently, it theoretically can produce absolute purity. Practically, however, it is limited by the control of the temperature of the distillation unit, the time involved in processing, and the bulk of the processing unit. The large bottle gas companies are able to handle the process and they do a remarkable job in purifying gases regularly[9].
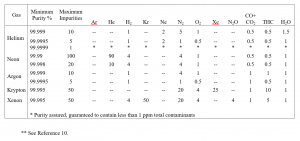
Unfortunately, the purification process is expensive, as reflected in the price of the bottled specialty gas. The bottled gas is certainly convenient. The efficiency, which is the ratio of the number of impurities removed relative to the total number of impurities arriving per unit time, is zero for bottled gases. Table II shows an example of the purity of some of the specialty inert gases now bottled[10]. More shall be said later about the “bottled” gas purity.
The second process, diffusion, has a limited use. Diffusion through a wall-like heated quartz is strongly enhanced for light mass gases[11]. Consequently, the process is limited to the lightest inert gas, helium, as long as hydrogen is removed.
The cost of this process is low with little maintenance cost. The process is inconvenient since it is limited to only helium and has very low flow rates for the purified gas. It is reasonably efficient. The purity of the gas is limited by the original hydrogen or water vapor in the gas, since hydrogen will diffuse through as rapidly as the helium.
The third purification process, chem-adsorption, is simply, the chemical reaction of a gas with the surface of a solid. It is enhanced by diffusion of the gas into the solid and the porosity of the solid. Chem-adsorption is separated into two traditional classes: the so-called phys-adsorption process, where the chemical bond of the gas to the walls of the adsorbent surface is weak, and regular chem-adsorption[12].
The phys-adsorption process[13] explains the effectiveness of xeolite, activated charcoal, and other, so-called molecular sieve materials[14, 15]. These compounds are characterized by high porosity and long channels due to an open crystal lattice structure. These porous compounds provide a large surface area for the weak chemical bond of the reactive gas.
TABLE II
TYPICAL PURITIES OF BOTTLED INERT SPECIALTY GASES**
Because of the weak chemical bond, the phys-adsorb material must be cooled to cryogenic temperatures to improve its adsorptive power. Consequently, inert gases are often “pumped” as well because the gas liquefies on the surface of the material. Once the inert gas is “pumped,” the efficiency to remove reactive gases is lowered.
In addition, the weak chemical bond to the gas allows the phys-adsorb material to be “reversibly” outgassed at elevated temperatures in a high vacuum. The outgassing is just the reversal of the chemical reaction by lowering the partial pressure of the gas. It is just this “reversibility” that causes another limitation to the material. Unless an ultra-high vacuum (less than 10-9torr) out-gassing is performed at the maximum absolute temperature difference from the operating temperature, the vapor pressure of the remaining reactive gases can actually be a contaminate to the purification process. A phys-adsorber cannot, in general, be exposed to air at room temperature and maintain its efficiency.
The phys-adsorption process is, in the final analysis, a variant on the fractional distillation process requiring ultra-high vacuums, high temperatures, and cryogenic temperatures. It is less bulky than fractional distillation, but has lower purity with increasing operating temperature. It can be used with best efficiency, cost, and convenience for helium gas purification with the operating temperature just above the boiling point of helium.
The cost of the phys-adsorb purifier is low, but the related cryogenic equipment, heaters, and ultra-high vacuum system cost is quite high. However, the maintenance cost is low. The process is convenient only for helium gas. Other inert gases require special cryogenic facilities to keep from liquefying the inert gas while removing the reactive gases. The efficiency is variable depending on the amount of inert gas liquefied and the total number of remaining sites for phys-adsorption.
Finally, there is the regular chem-adsorption or gettering process where a strong chemical bond is formed on the surface of the solid[16].
The major success and drawback of the chem-adsorption process is that it is specific to different reactive gases. Each chem-adsorbent reacts well with some gases and not well with others. Furthermore, it is more “reversible” for some gases than others. However, since a strong chemical bond is formed, one can expect a high efficiency because the reaction conversion factor, (the ratio of impurity atoms removed to chem-adsorber atoms), is on the order of 1.0.
The term coined to denote the end of the useful life of a chem-adsorber is the breakthrough point[17]. It is usually measured at the point at which the impurity pressure rises above certain tolerable limits. The point scales with the total number of chem-adsorber atoms in integral fashion dependent upon the geometry of the chem-adsorbing chamber. Because of the large reaction conversion factor, the break-through point can be quite large. Consequently, for a small amount of chem-adsorber, any one of the related requirements — long time between replacement, ultra-high purity, or large flows — can be obtained.
The more commonly used chem-adsorbers are:
1. The alkali metals,
2. Uranium,
3. Copper,
4. Titanium and other transition metals.
(1) The alkali metals, including calcium, have traditionally been used in glove boxes. They react at room temperature, are hard to handle, and react principally with O2, H2O, CO, and CO2. Calcium can react with N2.
(2) Uranium is an excellent chem-adsorber for impurities[5, 18, 19]. However, it is difficult to handle once activated, because it reacts at room temperature. It is also expensive, and sometimes hard to obtain.
(3) The Cu deposited on diatomaceous earth (B.T.S.) is very useful when operated at 100°C in two columns with a cold trap[20]. One column is reduced to Cu (B.T.S.) and the other is oxidized CuO. This combination picks up H2, O2, CO, and hydrocarbons, and converts them to H2O and CO2, which is removed by a liquid nitrogen cold trap. However, it does not remove N2 and can exothermically react if suddenly exposed to air at room temperatures.
(4) Finally, there are the transition metals like titanium[5, 17], zirconium, and palladium[16]. The most versatile and easy to obtain is titanium. It can be considered in detail. Titanium can be obtained at spectroscopic quality in a sponge form with a large surface area. It does not appreciably react at room temperature, but at 900°C has excellent chem-adsorption properties for N2 as well as the other reactive gases[5].
The chem-adsorption properties can easily be tested by observing that titanium is used as a gettering device or sublimation pump in a high vacuum system. The titanium sublimation pump has relative pumping speeds that are related to the chem-adsorption properties of Ti to gas impurities.
In addition, the work of M.L. Kyle, L. F. Coleman, R. D. Pierce, and J. P. Arntzen[5] show that titanium sponges can be reacted with a conversion factor N-atoms/Ti-atoms, of 0.32 at a gas concentration of 50 ppm to 0.68 at a gas concentration of 50,000 ppm of nitrogen impurities in argon.
Other workers[21] have found that by taking special precautions, the titanium chem-adsorber can reduce the oxygen impurity level to 1 part in 108.
The cost of a Ti chem-adsorber is low including a simple 900°C furnace[17]. The maintenance cost is small and certainly competitive with the cost of bottled gas. The process is convenient and useful for all the inert gases. The efficiency is high, as long as the flow rate is within tolerance. The ultimate purity is limited again by the flow rate.
It would appear that there is no simple answer to the question of how to purify inert gases. However, the most convenient, efficient, and low cost purifier for all the gases is the titanium chem-adsorber. In the next section, one such purifier unit is described, along with the important elements to maintain high purity.
In this section the practical problem of coupling purified gas or gas purifier to its point of use will be considered. Purity is unfortunately related to difficulty and, after 99.9% purity is reached, a number of related procedures must be performed to maintain higher purity. It is the object of this section to reduce the difficulty to a minimum.
Examine the whole purification system: a source of inert gas, a purifier, connecting lines, a gas purity monitor, and the process system. The system should be reasonably independent of the initial purity, have maximum final purity, adequate flow, long time maintenance purity, low or moderate cost for initial investment, low cost for maintenance, minimum bulk, and be easy to handle.
One possible solution appears to be using ultra-high purity cylinder gas and removinge the purifier. However, this possibility can be more expensive to maintain in comparison to the intermediate purifier. In addition, the high purity bottled gas can easily lose purity. A partially opened valve or a diffusive leak can contaminate the bottle. Also, the bottle outgases, no matter how well constructed and processed, because of thermal cycling of the walls[22]. In general, it is difficult to maintain bottled gas purity greater than 99.9999% over a period of time.
Once a purifier is considered, one can quickly see from the previous section that the titanium chem-adsorption system has the convenience, efficiency, and low cost necessary for a general purpose unit. With proper design, adequate flow rates can be obtained at any given final purity requirement. The initial cost can be maintained at a moderate level and the maintenance can be kept low, as long as, standard inert gas cylinders (~99. 9% or greater) are used. Such a unit can be seen in the Figure 1, where the size of the unit is about 7″ high x 19″ wide x 10″ deep.
Figure 1
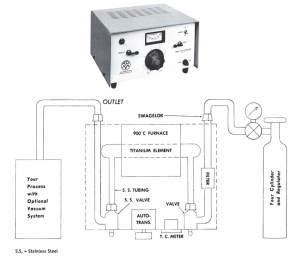
GP-100 INERT GAS PURIFIER
Particle Filter: On inlet side 7 microns nominal, replaceable
Titanium Element: Quartz, replaceable
Furnace: 400 watts maximum, 0-1000° C
Temperature Control: Chromel – Alumel Thermocouple, meter on front panel along with autotransformer control
Outlet: All stainless steel fittings, toggle valve, 1/4” Swagelok coupling
Inlet: 1/4” Swagelok coupling, particle filter and 1/4” toggle valve
Maximum Flow: At 99.9% pure inlet gas 1/4 liter/min. (0.53 cu. ft. /hr.) at atmosphere pressure
Pressure-Vacuum: 10 psi maximum, helium leak tight vacuum
Power Supply: 110VAC, 5A; switch, pilot light and fuse on front panel
Size: 7-3/16” H x 19-1/8” W x 10-1/2” D
Weight: 20 Pounds
Cooling: Requires 1” clearance on all sides for adequate air cooling
Options: Also available in 220VAC unit, or all Stainless Steel tubing and parts throughout gas flow path
The breakthrough point for nitrogen is roughly 300 cu. ft. of 99.9% purity gas. The unit can purify 99.8% pure argon in a worst case test to 99.9991%, with impurities like H2O-2 ppm, N2/CO-3ppm, O2-2 ppm, CO2-1 ppm, Ethane 1 ppm, Hydrocarbons 0-ppm, with the limits of detectability set at 1 ppm within a 24 hour period[23]. The unit can deliver up to 1/4 liter/min flow at 99.999% purity. Since most gas cylinders start with purities in excess of 99.8%, the titanium unit will generally deliver purities in excess of 99.9999% and can maintain purity for more than one bottle before changing elements.
So far it has been assumed that the only contaminants come from gas cylinders. However, this is generally not the case. The major sources of contamination in all gas purification systems are leaks. It is of primary importance to leak check the system from the gas bottle to the process end of the line. A helium gas analyzer leak detector is preferred for this purpose.
Another source of contamination is outgassing and impurity diffusion through the walls of the gas lines. All glass or stainless steel systems are preferred with copper used in the low purity side. If possible, every part of the gas line should be flamed or baked out at 200°C to remove impurities from the walls. This outgassing process must be done in a vacuum or with purging inert gas and must be repeated every time the gas line is opened to the air. If quick connections are necessary, Viton-A or Neoprene should be used for the seals. Avoid tubing like PVC, latex, tygon, etc., because they are all porous to either H2O or CO2.
There are a number of gas purity monitors commercially available for O2 and H2O detection above 100 ppm impurity (99.99%). Recently, two versions of Ti or W filament detectors have been considered[24, 25]. These devices detect the partial pressure of gases that react with the filament, usually O2, H2O, CO2, and hydrocarbons. For the Ti gas purifier, impurities O2 and H2O have the smallest breakthrough point and reduce the breakthrough point of N2. Consequently, these monitors are excellent for determining the useful life of the purifier.
Finally, the system to be used for processing must be leak tight, outgassed, and made of stainless steel or glass. The escape of the used inert gas from the system must be just as carefully taken care of. The system may be either closed and leak tight, the outlet port evacuated with a fore pump and cold trap or the outlet line passed through a cold trapped bubbler. For ultra-high gas purities, it is just as important to remove impurities that may back diffuse through the outlet as to remove those that diffuse into the inlet.
There are six inert gases — He, Ne, Ar, Kr, Xe, and Rn. He and Ar are the most commonly available and the most often used. While no single purification process solves all problems, a titanium chem-adsorption process has a low cost, is convenient and efficient, and can produce ultra-high purities of all the inert gases. Other purification techniques are mentioned.
A gas purification system consisting of a gas cylinder, purifier, gas lines, gas monitor, and process system is analyzed. A titanium chem-adsorber purifier is described.
Precautions for obtaining an ultra-high purity system are as follows:
a) Leak check the whole system
b) Use only stainless steel and glass with copper on low purity side
c) Outgas lines and process system
d) If quick connections are required, use only Viton-A or Neoprene and keep them cool
e) Use a gas monitor for measuring the impurity with the smallest breakthrough point
f) The outlet line must isolate the system from back diffusion into the process system.
The author is indebted to Worth Allred, Dr. James Whelan and Dr. Eddie Kroeger of USC; Allen Cox of Gardner Cryogenics; Fred Schrey of Hughes; Ken Lehner of AIRCO; and Dr. Hellmut Fritzsch of the University of Chicago, for several important conversations. He acknowledges with pleasure the analysis work of Dr. Dwight Fisher of West Coast Technical Service, literature documentation by A. F. Tolivar of WESRAC, the technical assistance of David E. Moore, and the assistance of RD Mathis Company.
Author
R. T. Payne, Ph. D.
Distributed By
RD Mathis Company
2840 Gundry Ave Signal Hill CA 90755
Phone: (562) 426-7049 | Fax: (562) 595-0907
1) Handbook of Chemistry and Physics, 47th Edition, Robert C. Weast, Editor, (The Chemical Rubber Co., Cleveland, 1966)
2) The chemical reactivity of the heavy inert gases Xe and Kr is the subject of intense research. The compounds formed are oxidizers and fluoridizers. They are extremely unstable and do not form as long as there are competing reactions for fluorine and oxygen. Consequently, xenon and krypton can be considered inert for all practical purposes.
3) Bartlett, Neil, Dr., “What Now Noble Gases?”, Industrial Research II, #5 page 70 (May, 1969)
a. XeF2, XeF4, (Xe06)4- Xe04, Xe03, Xe + KF2
b. All available information indicates no Ar, or lighter. Low activator energy used as oxygizer for fluoridizers. Important not to break activated chem-adsorber.
4) Gray, Dwight E., Ph.D., American Institute of Physics Handbook, Second Edition, (McGraw-Hill Book Company, New York, 1963)
5) Kyle, M. L., Coleman, L. F., Pierce, R. D., and Arntzen, J. D., “Kinetics of the Titanium-Nitrogen Reaction with Application to the Design of Gettering Systems” Argonne (ANL-7167) cost $3.00 Obtain the article from the Clearing House for Federal Scientific and Technical Information, Springfield, Virginia 22151.
6) White, P. A. F., and Smith, S. E., “Inert Atmospheres” (Butterworths, Washington, D. C. 1962)
7) Walton, G. N., Ed. “Gloveboxes and Shielded Cells” (Butterworths, London, 1958)
8) Kohl, A. L., and Riesenfeld, F. C., “Gas Purification” (McGraw-Hill, New York, 1960)
9) Information about inert gases may be obtained from Air Reduction, Inc. (AIRCO), Rare and Specialty Gas Dept., Union Landing Road, Riverton, New Jersey 08077; Gardner Cryogenics Corp., 2136 City Line Rd., Lehigh Valley Industrial Park, Bethlehem, Penn. 18001; and a number of other gas suppliers.
10) Taken from the AIRCO, Rare and Specialty Gases Catalog, copywright 1963 Page 6. Air Reduction, Inc. 150 East 42nd St., New York, N. Y. 10017 Other gas companies have similar purity analysis.
11) Dushman, Saul, “Scientific Foundations of Vacuum Technique” Pages 491-500 (John Wiley & Sons, New York, 1962)
12) Refer to Reference 11, see page 376.
13) Refer to Reference 11, Chapters 6 and 7.
14) Trade name of product from the Linde Division, Union Carbide Corp.
15) King, James, Jr., and Benson, Sidney W., “Electrostatic Interactions in Gas-Solid Chromatography”, Analytic Chemistry 38, Pgs. 261 – 265 (1966)
16) Refer to Reference 11, Chapter 8.
17) Kyle, M. L., Arntzen, J. D., and Coleman, L. F., “Design and Operation of a Pilot-Plant Gettering System of Titanium-Metal Sponge for Removal of Nitrogen from Argon”. Argonne (ANL-7128) Cost S2. 00 – Obtain the article from the Clearing House for Federal Scientific and Technical Information, Springfield, Virginia 22151.
18) Porto, S. P.S., private communication. Uranium is used as a getter for He – Ne lasers.
19) Smith, Maynard E., Hansel, James M., and Waterbury, Glenn R., “An Inert Atmosphere Enclosure for the Preparation of Samples Prior to the Determination of Oxygen”. Los Alamos (LA-3344) Cost $1.00 Obtain the article from the Clearing House for Federal Scientific and Technical Information, Springfield, Virginia 22151.
20) Maak, F., and Sellars, C. M., “A Simple Method of Inert Gas Purification”, J. Sci. Instrum. 42, pg. 900 (1965)
21) Reed, T. B., private communication. The limiting partial pressure calculated for oxygen impurities on Titanium is 10-11torr. See A. Mitchell, Nature 201, 390 (1964)
22) Cox, Al, private communication.
23) The impurity levels were measured with a mass spectrometer by Dr. Dwight Fisher of West Coast Technical Service.
24) McKee, J., “An Inert Gas Purity Tester”, United Nuclear Corp. White Plains, New York
25) Reed, T.B., and LaFleur, W. J., “Measurement of Trace Oxygen and Water Vapor in Inert Gases” Solid State Research Report, 3 Pg. 27 (1967) Lincoln Laboratories.
